Law
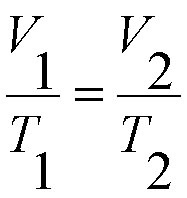 charle´s law formula
charle´s law formula
Charles' Law
For a fixed mass of gas at constant pressure, the volume is directly proportional to the kelvin temperature.
That means, for example, that if you double the kelvin temperature from, say to 200 K to 400 K, at constant pressure, the volume of a fixed mass of the gas will double as well.
Mathematically it can be expressed as: V = constant x T
When you heat up a gas, but not the air outside you can see a demonstration of the law. This happens in hot air balloons.
Gas molecules will now be moving faster, and so will hit the barrier more frequently, and harder. The air molecules on the outside are hitting it the same way as before.
Obviously, the barrier will be forced to the right, and the volume of the gas will increase. That happens until the pressure inside and outside is the same.
So to review, with a fixed mass of gas, the pressure is the same before and after. And the volume will increase when you increase the temperature of the gas.
NOTE: temperature in charles law is in kelvins a way to change it to celsius is the following:
ºc+273=K
http://www.chemguide.co.uk/physical/kt/otherlaws.html
For a fixed mass of gas at constant pressure, the volume is directly proportional to the kelvin temperature.
That means, for example, that if you double the kelvin temperature from, say to 200 K to 400 K, at constant pressure, the volume of a fixed mass of the gas will double as well.
Mathematically it can be expressed as: V = constant x T
When you heat up a gas, but not the air outside you can see a demonstration of the law. This happens in hot air balloons.
Gas molecules will now be moving faster, and so will hit the barrier more frequently, and harder. The air molecules on the outside are hitting it the same way as before.
Obviously, the barrier will be forced to the right, and the volume of the gas will increase. That happens until the pressure inside and outside is the same.
So to review, with a fixed mass of gas, the pressure is the same before and after. And the volume will increase when you increase the temperature of the gas.
NOTE: temperature in charles law is in kelvins a way to change it to celsius is the following:
ºc+273=K
http://www.chemguide.co.uk/physical/kt/otherlaws.html
Application in real life

It is really significant for many people such as scuba divers since they depend on knowing the temperature of the surface or deep under the sea.
Also, Charles Law is used when tires get overheated on hot summer days and they explode. This happens because temperature make the tires volume increase so much that it can not be handle and that is the reason of the explosion
Also, Charles Law is used when tires get overheated on hot summer days and they explode. This happens because temperature make the tires volume increase so much that it can not be handle and that is the reason of the explosion
HYdrogen balloon

Jacques Charles contributed with his invention of the first hydrogen balloon in 1783. Charles and Nicolas Robert made the first to ascend on a gas hydrogen balloon on December 1, 1783. After that, he use his knowledge in hydrogen with Nicolas Robert's new method of coating silk with rubber. This helped create the first hydrogen balloon.
http://inventors.about.com/od/astartinventions/ss/airship_3.htm
http://inventors.about.com/od/astartinventions/ss/airship_3.htm
The megascope

Charles invented the megascope in 1780. It is an optical instrument for projecting the magnified image of any object. This let him and other scientists present their experiments on a bigger scale.
He also used the solar microscope to advantage in his optical lecture, that he gave on summers when the sunlight let it.
http://books.google.com.mx/books?id=O9e_7E22caAC&pg=PA61&lpg=PA61&dq=jacques+charles+megascope&source=bl&ots=q3N6VXT9z0&sig=SPwO6mA6aiLdvZz3gCDcIb80ObI&hl=es&sa=X&ei=H68GU832H6eG2wW1o4H4CQ&ved=0CEoQ6AEwAw#v=onepage&q=jacques%20charles%20megascope&f=false
He also used the solar microscope to advantage in his optical lecture, that he gave on summers when the sunlight let it.
http://books.google.com.mx/books?id=O9e_7E22caAC&pg=PA61&lpg=PA61&dq=jacques+charles+megascope&source=bl&ots=q3N6VXT9z0&sig=SPwO6mA6aiLdvZz3gCDcIb80ObI&hl=es&sa=X&ei=H68GU832H6eG2wW1o4H4CQ&ved=0CEoQ6AEwAw#v=onepage&q=jacques%20charles%20megascope&f=false
